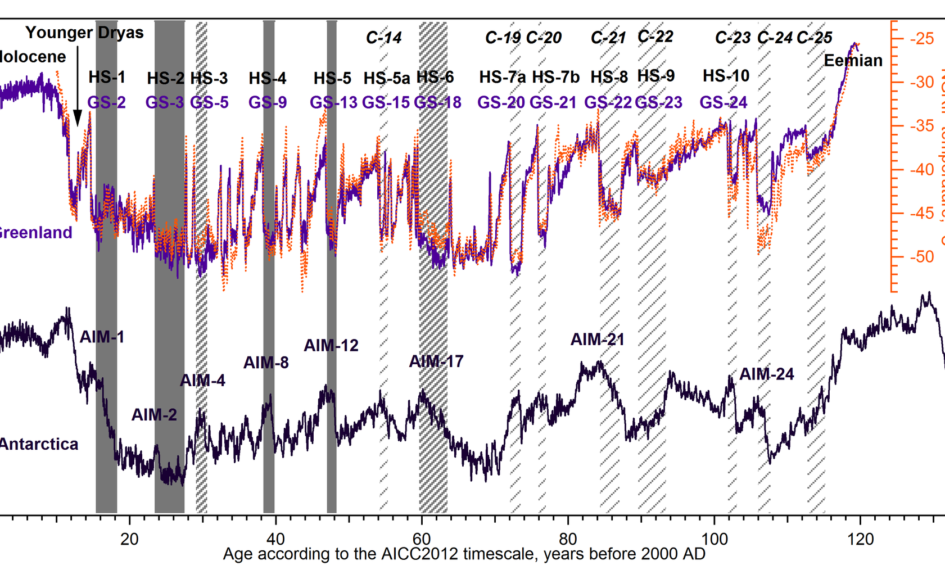
Meaning of Isotope: Isotopes are variants of a particular chemical element that have the same number of protons in their atomic nuclei but differ in the number of neutrons. This difference results in varying atomic masses for the isotopes of an element. Despite these differences, isotopes of an element exhibit very similar chemical behaviors. Isotopes are commonly used in various scientific fields, including chemistry, physics, medicine, and environmental science.
Etymology and Origin: The term “isotope” comes from the Greek words “isos,” meaning “equal,” and “topos,” meaning “place.” It was coined by the British chemist Frederick Soddy in 1913.
- Greek Roots: The Greek roots signify “same place,” referring to the fact that isotopes of an element occupy the same position on the periodic table. Although they have different atomic masses, their chemical properties are similar because they have the same number of protons and electrons.
- Scientific Adoption: Soddy introduced the term “isotope” to describe the phenomenon he and others had observed where elements appeared in different forms with different atomic weights, yet seemed to occupy the same position (or “place”) in the periodic system of elements.
Thus, “isotope” combines the Greek words for “equal” and “place” to describe different forms of the same element that share chemical properties but have different atomic masses.